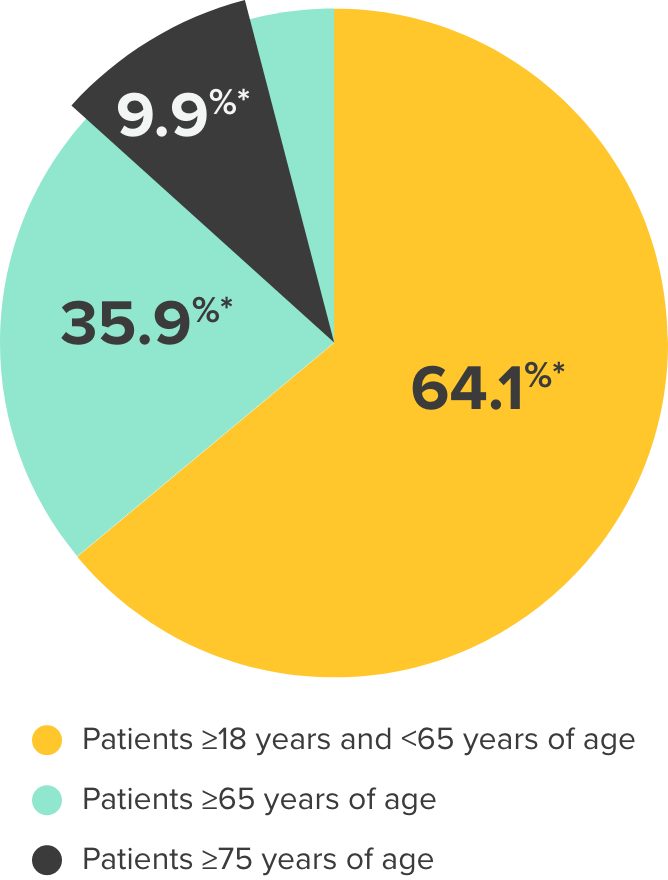
MYRBETRIQ is contraindicated in patients with known hypersensitivity reactions to mirabegron or any inactive ingredients of the tablet.
MYRBETRIQ monotherapy or in combination with solifenacin succinate can increase blood pressure in adults. Periodic blood pressure determinations are recommended, especially in hypertensive patients. MYRBETRIQ is not recommended for use in patients with severe uncontrolled hypertension (defined as systolic blood pressure ≥ 180mm Hg and/or diastolic blood pressure ≥ 110mm Hg). Worsening of pre-existing hypertension was reported infrequently in patients taking MYRBETRIQ.
In patients taking MYRBETRIQ, urinary retention has been reported in patients with bladder outlet obstruction (BOO) and in patients taking muscarinic antagonist medications for the treatment of OAB. A controlled clinical safety study in patients with BOO did not demonstrate increased urinary retention in patients treated with mirabegron; however, MYRBETRIQ should still be administered with caution to patients with clinically significant BOO. For example, monitor these patients for signs and symptoms of urinary retention. MYRBETRIQ should also be administered with caution to patients taking muscarinic antagonist medications for the treatment of OAB, including solifenacin succinate.
Angioedema of the face, lips, tongue and/or larynx has been reported with MYRBETRIQ. In some cases, angioedema occurred after the first dose. Cases have been reported to occur hours after the first dose or after multiple doses. Angioedema, associated with upper airway swelling, may be life threatening. If involvement of the tongue, hypopharynx, or larynx occurs, promptly discontinue MYRBETRIQ and initiate appropriate therapy and/or measures necessary to ensure a patent airway.
Since MYRBETRIQ is a moderate CYP2D6 inhibitor, the systemic exposure to CYP2D6 substrates is increased when co‐administered with MYRBETRIQ. Therefore, appropriate monitoring and dose adjustment may be necessary, especially with narrow therapeutic index drugs metabolized by CYP2D6.
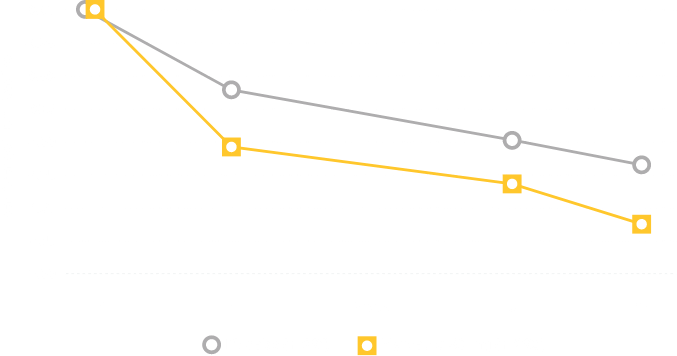
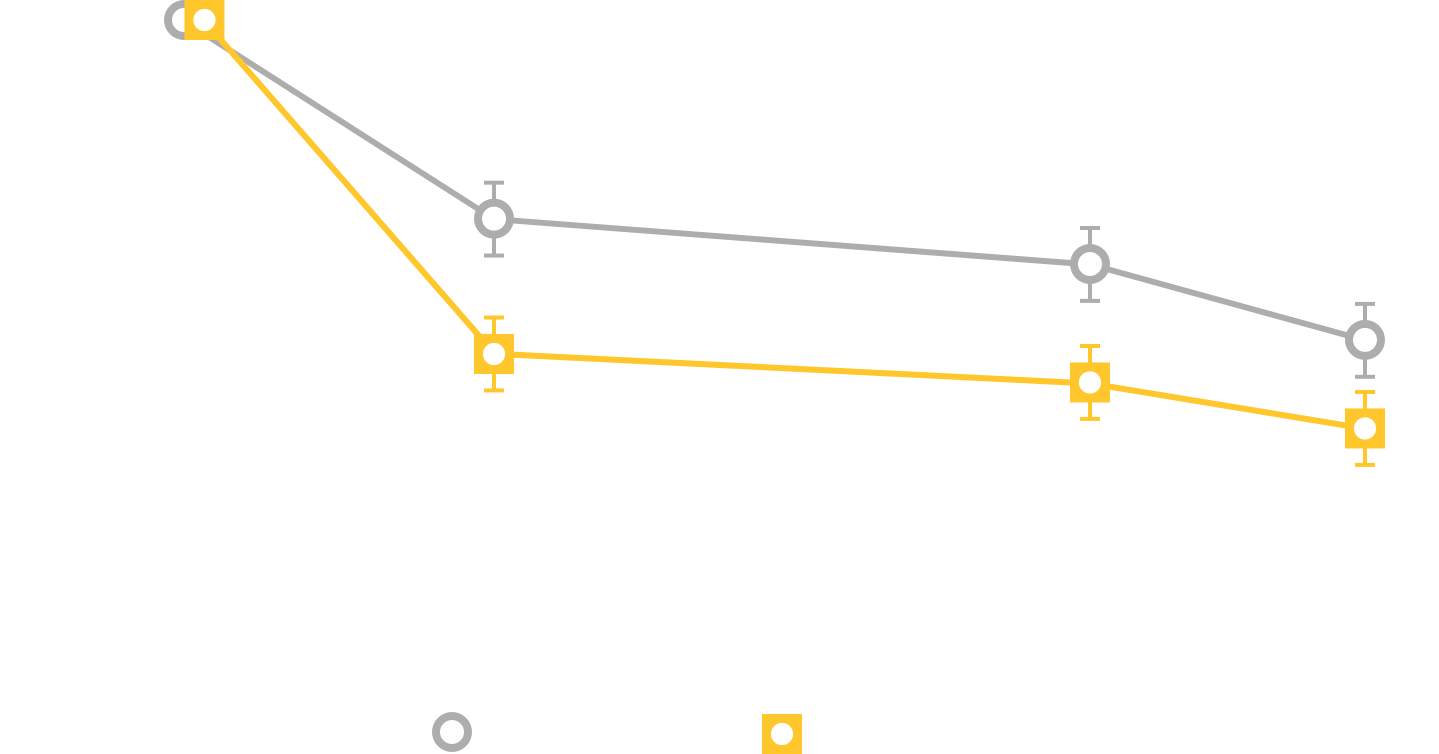
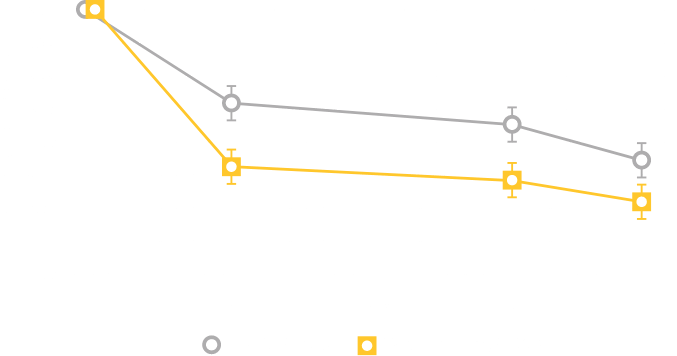
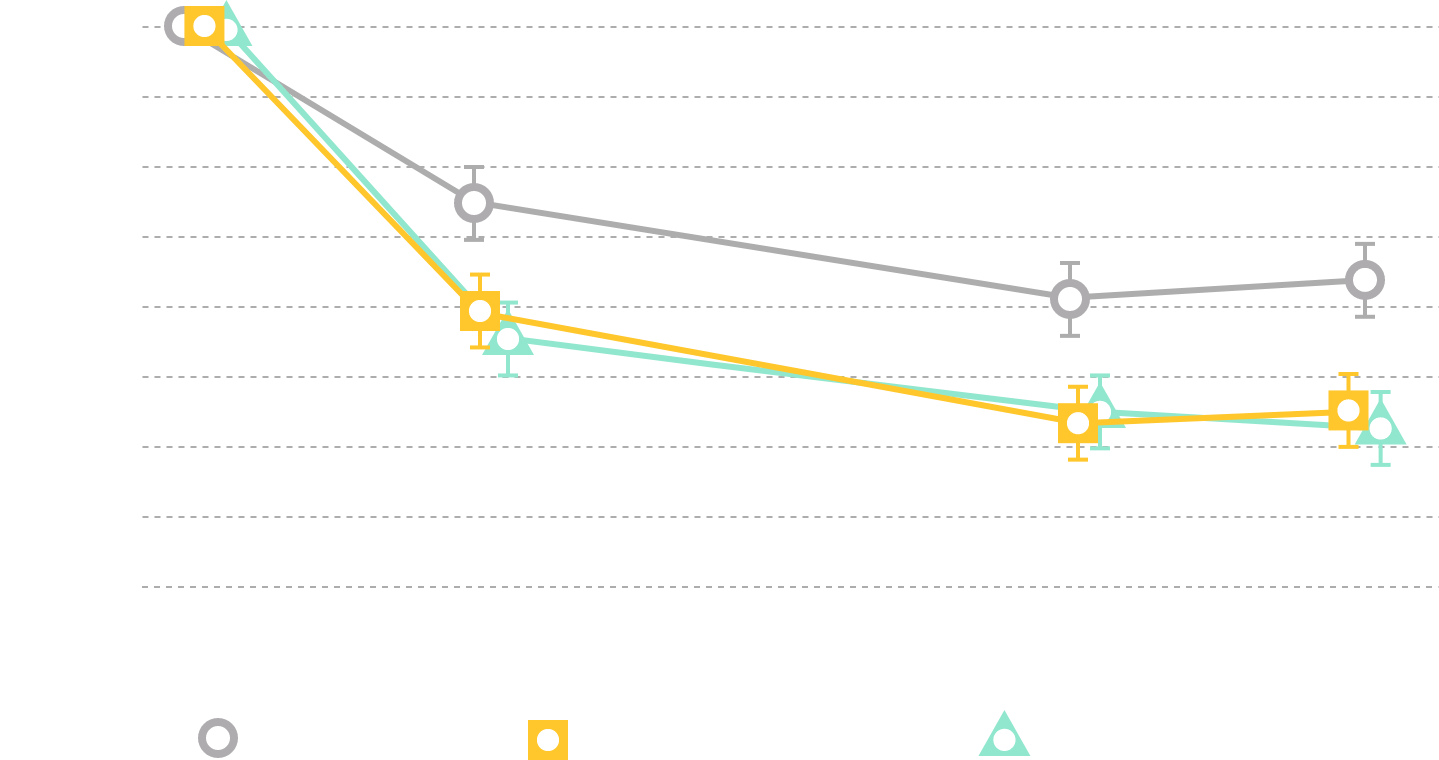
In clinical trials, the most commonly reported adverse reactions in adults (> 2% and > placebo) for MYRBETRIQ 25mg and 50mg versus placebo, respectively, were hypertension (11.3%, 7.5% vs. 7.6%), nasopharyngitis (3.5%, 3.9% vs. 2.5%), urinary tract infection (4.2%, 2.9% vs. 1.8%), and headache (2.1%, 3.2% vs. 3.0%).
In clinical trials, the most commonly reported adverse reactions in adults (> 2% and > placebo and > comparator) for MYRBETRIQ in combination with solifenacin succinate 25mg + 5mg and 50mg + 5mg versus MYRBETRIQ 25mg, MYRBETRIQ 50mg, solifenacin succinate 5mg, and placebo, respectively, were dry mouth (9.3%, 7.2% vs. 3.8%, 3.6%, 6.5%, 2.2%), urinary tract infection (7.0%, 4.0% vs. 4.0%, 4.2%, 3.6%, 5.3%), constipation (4.2%, 3.9% vs. 1.2%, 2.8%, 2.4%, 1.2%), and tachycardia (2.2%, 0.9% vs. 1.6%, 1.6%, 0.7%, 0.8%).
In postmarketing experience with mirabegron, the following events have also occurred: atrial fibrillation, nausea, diarrhea, and dizziness.
Please refer to prescribing information for solifenacin succinate when prescribing MYRBETRIQ in combination with solifenacin succinate.
Please click here for complete Prescribing Information for MYRBETRIQ® (mirabegron extended-release tablets)